From Well Plates to Cartridges: How CaDI Reimagines the ELISA Workflow
Immunoassays have remained the backbone of protein detection across research and clinical diagnostics for decades. Among them, a widely used method known as enzyme-linked immunosorbent assays (ELISAs) serves as the gold standard for protein detection, prized for its versatility, high sensitivity, and quantitative capabilities. However, as lab demands shift toward faster turnaround times and more efficient workflows, the limitations of traditional ELISAs have become increasingly apparent. ELISAs require labor-intensive workflows or costly robotic systems, multi-hour runtimes, dependency on trained users, and specialized equipment.
Emerging innovations like the capillary-driven immunoassay (CaDI) platform are redefining the possibility for high-sensitivity, quantitative immunoassays beyond the laboratory. To understand what that really means in practice, the following sections walk through how an ELISA works, how CaDI reimagines the ELISA workflow, and how these approaches support different applications and assay development needs.
What is ELISA exactly? A Powerful but Workflow-Heavy Method
An ELISA is a type of laboratory immunoassay used to detect and quantify specific proteins such as antibodies, antigens, or biomarkers across various sample types and applications. At a high level, it works by using antibodies that bind to a specific target. When the target is present in a sample, it binds to these antibodies, triggering a chemical reaction that produces a measurable signal.
The resulting signal, often a color change or light emission, is directly proportional to the amount of target present in the sample. By comparing the signal in an unknown sample to a set of known samples (i.e., standards), researchers can determine the precise concentration of the target in a sample. ELISA emerged as the gold standard by combining three powerful advantages: 1) high specificity, 2) elevated sensitivity, and 3) quantitative results.
However, achieving this level of performance requires tradeoffs in efficiency. A traditional ELISA requires many manual steps, long incubation times, precise timing, careful reagent addition, and often requires 4+ hours to complete at a minimum. Additionally, each step must be executed with precision to maintain the quality of the data. Due to the complexity, ELISAs cannot be feasibly run outside of laboratory settings. These complexities are precisely what CaDI was designed to address.
What is CaDI? A Simplified, Rapid ELISA
Capillary flow-driven immunoassay (CaDI) is a simple, cartridge-based device that streamlines the numerous manual ELISA steps and performs them automatically. Instead of requiring repeated pipetting and incubation steps, CaDI handles the entire ELISA process inside a single microfluidic cartridge.
CaDI streamlines each step of an ELISA, requiring only a single sample addition from the user. Once the sample is introduced, the device uses capillary-driven flow to enact a series of automatic steps, including target binding to the capture antibodies, washing away unbound material, detection antibody delivery, substrate delivery, and chemiluminescent signal generation.
The reagents required for the assay are pre-loaded onto reagent pads within CaDI’s microfluidic channels. As the assay runs, these reagents are rehydrated and delivered to the detection zone in a controlled sequence. CaDI’s precise channel geometry governs the timing of each step, ensuring the assay proceeds in the correct order without requiring user intervention.
By replacing a multi-hour workflow with a fully automated process, CaDI delivers the same high-quality, quantitative results as an ELISA in just 10 minutes, making it more than 25 times faster and less complex to run outside of a traditional laboratory.
Immunoassays are Evolving: A Step-by-Step Look at ELISA v. CaDI
To better understand how these platforms differ in practice, the following section walks through each step of a traditional ELISA and highlights how CaDI simplifies the same process.
Pre-Assay Preparation: Capture Antibody Coating and Blocking
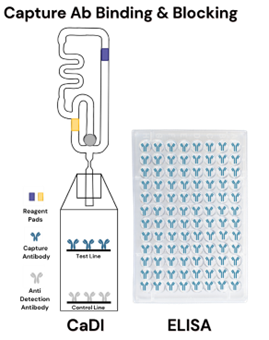
In a typical ELISA workflow, users begin with a pre-coated and pre-blocked 96-well plate in which capture antibodies have already been immobilized onto the plate surface during manufacturing. In addition to immobilizing the antibodies, the plate surface is treated with blocking agents to prevent non-specific binding during the assay. Non-specific binding occurs when molecules other than the intended target unintentionally attach to the antibodies. When this occurs, the background signal increases, thereby reducing assay sensitivity. From the user’s perspective, these plate preparation steps are complete before the assay begins.
Likewise, in CaDI, capture antibodies are immobilized during manufacturing. However, instead of being bound to polystyrene wells, the antibodies are coated onto a membrane within the cartridge through a validated manufacturing process to ensure consistent performance and stability. Blocking is also integrated directly into the cartridge. Membrane-based blocking is applied during manufacturing, and additional blocking components are incorporated into the assay buffer to help prevent unintended molecular interactions within the device.
Both ELISA and CaDI therefore address capture antibody immobilization and background signal minimization prior to assay initiation, differing primarily in structural format rather than underlying function.
Step 1: Sample Addition
In a plate-based ELISA, each sample must be added individually to separate wells. This repetitive step requires careful precision and potentially increases the risk of pipetting errors or cross-contamination between wells. When running many samples, this step alone can account for a significant amount of hands-on time.
CaDI cartridges only require a single sample addition step that’s simple to execute. Instead of pipetting samples into wells, the user adds the sample to the cartridge using a simple dropper tube. From there, the cartridge automatically disperses the sample through the CaDI device using capillary-driven flow.
Step 2: Detection Antibody Binding
In an ELISA, detection antibodies are added manually using a pipette. This step is critical because these enzyme-linked detection antibodies must bind to the target molecule to generate a measurable signal. After the antibodies are added, an incubation period is required to allow sufficient time for binding to occur.
In CaDI, the detection antibody is stored on a reagent pad within the cartridge and is automatically released after the target has bound to the capture antibodies. This controlled timing supports consistent binding and downstream signal development without manual pipetting or additional user steps.
Step 3: Wash
Wash steps are arguably one of the most labor-intensive and critical aspects of ELISA. Washing plays an important role in ELISA by removing unbound molecules, which helps maintain a low background signal. A typical ELISA involves multiple wash cycles, often three or more washes per round across several stages throughout the assay. These washes are performed manually or with a plate washer, and consistency depends on user technique or instrument calibration.
In CaDI, washing is integrated directly into the cartridge. After the detection antibody has been delivered to the detection zone, a wash buffer “plug” stored in the right channel flows through the detection zone to remove any unbound molecules. This automated wash step helps maintain a low background signal and consistent performance without requiring additional user steps.
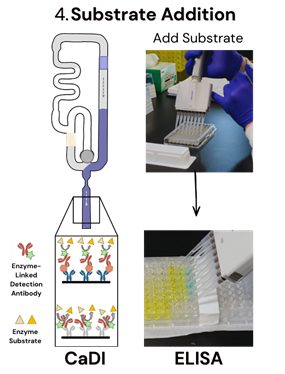
Step 4: Substrate Addition
In an ELISA, the substrate is added after the detection antibody has bound to the target and unbound molecules have been washed away. The substrate then reacts with the enzyme linked to the detection antibody to produce a measurable signal. To control signal development, a separate stop reagent is added after a defined period, and the signal must be read within a specific window to ensure accurate results. These sequential steps require very careful timing to control signal development and measurement.
In CaDI, a reagent pad containing the substrate is pre-loaded within the cartridge and delivered automatically at the appropriate point in the assay. Once released, it flows to the detection zone without user intervention, ensuring consistent timing and reducing any timing variability associated with manual handling.
Step 5: Chemiluminescent Detection
The final step of an ELISA involves measuring the chemiluminescent signal to quantify the target present in the sample. In a traditional ELISA workflow, this typically involves transferring the plate to a dedicated plate reader and managing data collection. Afterward, users must apply data analysis methods, such as curve fitting, to convert raw signal values into quantitative results.
In CaDI, chemiluminescent detection is performed using a compact, low-cost reader. The reader measures the signal emitted and automatically converts that signal into quantitative data. The integrated software also handles data analysis, automatically comparing the signal to a calibration curve to report a clear quantitative result. By combining sensitive chemiluminescent detection with automated data processing, CaDI delivers consistent, easy-to-interpret results in minutes.
Applications: Matching ELISA and CaDI to the Right Use Case
Both ELISA and CaDI are widely applicable immunoassay platforms. The difference mostly lies [GG4] in how and where they are best deployed.
ELISA Applications
ELISA continues to be a foundational tool across research and diagnostics. Its long history and broad adoption make it a strong choice for:
· Method development and early-stage exploration
· High-throughput testing workflows where extended incubation times are acceptable
· Applications that can accommodate longer assay runtimes
· Use cases designed for traditional laboratory settings
For teams working within traditional laboratory workflows, ELISA remains a reliable and well-understood approach to quantitative protein detection.
CaDI Applications
CaDI is built on the same immunochemical principles as ELISA, but delivers them in a faster, automated format. As a result, CaDI is well-suited for many of the same environments, while also expanding into new use cases. Common applications include:
· Point-of-need and decentralized testing environments
· Applications requiring rapid quantitative results
· Applications with limited lab infrastructure or staff
· Applications where small batch testing is required
By automating the ELISA workflow and reducing run time to approximately 10 minutes, CaDI allows researchers, clinicians, and other operators to focus more on decision-making rather than assay execution.
Custom Assay Development on the CaDI Platform
At Burst Diagnostics, we provide custom assay development to support unique partner needs. Our custom assay development services are designed for teams looking to translate ELISA-based assays into faster, automated formats or to build new assays on the CaDI platform from the ground up.
We work with partners to design and optimize assays tailored to specific analytes, sample matrices, and performance requirements, leveraging CaDI to deliver high-quality, rapid, quantitative results. Our technical expertise supports partners from initial concept through commercialization, allowing assays to quickly move beyond traditional formats and into applications that demand speed, simplicity, and reliability. Applications span veterinary diagnostics, clinical diagnostics, pharma, environmental monitoring, wellness, and other industry-specific use cases.
If you’re interested in custom assay development powered by the CaDI platform, visit our Custom Assay Development page or Contact Us to learn more.